
Vaccine Timeline
The timeline for the COVID-19 vaccine can be unclear to many, but if you want to look at the big picture, research began all the way back to the 1930s with the discovery of the first coronavirus infection in chickens. Decades later in 1965 Dr. June Almeida was able to identify a coronavirus under a microscope.

MRNA vaccine technology began in the 1990s. Katalin Karikó was on to the idea of using mRNA to have cells create specific proteins. Because of her determination she was able to make key discoveries and eventually partnered with Drew Weissman, M.D., Ph.D. in 1998 to study whether mRNA technology could be used for an HIV vaccine. Developing vaccines with mRNA continued in the U.S. because the National Institute of Health supported the research in the event of another flu pandemic like the one in 1918.
In 2003 the SARS epidemic and, a decade later, the identification of MERS prompted research on a vaccine for coronaviruses targeting the spike protein on the virus. This vaccine was developed in 2016, but it was hard to find funding and a way to test it when there was not a major outbreak at the time. This did not mean all research for this type of vaccine stopped, in fact, the lipids that are the packaging for the extremely delicate mRNA were approved by the FDA in 2018.

So, we see that years of research had already been established when COVID-19 was discovered in 2019. Scientists were able to isolate the virus causing COVID-19 and worked quickly to decode its DNA. It was not long before it was clear that this virus was causing a global pandemic. This is where things began to come together to create a vaccine in record time. The greatest scientists around the world prioritized and collaborated on research for a vaccine that had already had decades of work behind it. Scientists also didn’t have to wait for an agency to take the financial risk to fund it or to find people willing to participate in trials because concern about COVID-19 was so great. Many bureaucratic processes were avoided because of the urgency and in the U.S. the FDA used the same processes to evaluate the vaccines but were able to complete some parts at the same time given the number of resources available.

The vaccine is certainly something to be celebrated. Through hard-work, determination, and the global community working together, we have an incredibly effective way to protect ourselves from COVID-19 that’s available to everybody in our community. If you haven’t been vaccinated yet, here’s the place to start!
Vaccine Timeline
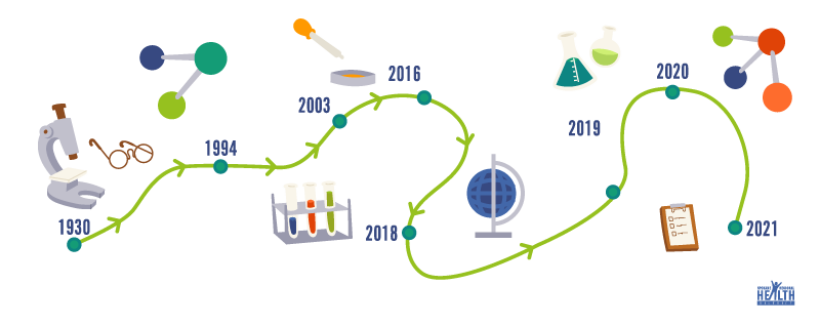
Click to view a printable, letter-sized Vaccine Timeline
